Clinical Evidence
Key Indications
Locoregional recurrent breast cancer
Breast cancer is the most common cancer in women worldwide and locoregional recurrence can occur in up to one-third of previously treated patients1. Treatment of locoregional breast cancer recurrences may involve surgery and radiation therapy or re-radiation therapy in patients who already underwent radiation therapy during initial treatment.
A recent metaanalysis analyzed eight two-arm studies including five randomized trials with a total of 627 patients who underwent radiation therapy alone or radiation therapy combined with superficial microwave based hyperthermia1. Patients were treated with a median of 7 hyperthermia sessions, attaining an average temperature of 42.5 °C. Mean radiation therapy dose was 38.2 Gy (range, 26-60 Gy). Hyperthermia was most frequently applied after radiation therapy. The complete response rate of the combined treatment was 60.2% being significantly improved as compared to 38.1% after radiation therapy alone (odds ratio 2.64, 95% confidence interval 1.66-4.18, P<.0001). The acute and late toxicity profile of the combined treatment was mild1.
Reference:
Locally advanced cervical cancer
Cervical cancer is the fourth most common cancer affecting women.
Long term results are available of a randomized trial comparing radiation therapy using a median total dose of 68 Gy (including a brachytherapy boost) and a total of 5 sessions of regional hyperthermia (42 °C for a 60-minute period) with radiation therapy alone in 114 patients with locally advanced cervical cancer1. The trial demonstrated sustained improvements in its primary endpoint being local control for the combined treatment (56% vs. 37%; p = 0.01) as well as improved overall survival after 12 years: 37% vs. 20% (p = 0.03) with similar grade 3 late toxicity events in both groups1.
For locally advanced cervical cancer combined chemoradiation is a standard of care. Less is known regarding whether hyperthermia is also able to improve cancer outcomes of chemoradiation. Thus, combination of hyperthermia and radiation therapy is recommended primarily for those patients unfit or not willing to undergo chemotherapy.
A recent network-metaanalysis of 59 randomized trials suggested equal effectiveness of combined hyperthermia and radiation therapy as compared to chemoradiation for locally advanced cervical cancer2.
References:
Pancreatic Cancer
Pancreatic cancer is one of the most common causes of cancer-related death worldwide. The only curative therapy option is surgical resection, which is applicable in only around one third of newly diagnosed patients. After resection adjuvant chemotherapy is indicated.
Results of the "Hyperthermia European Adjuvant Trial" (HEAT) are available, a randomized clinical trial which investigated regional hyperthermia (RHT) with cisplatin added to gemcitabine compared to chemotherapy with gemcitabine alone in the adjuvant treatment after resection of pancreatic cancer (1).
A total of 117 patients were randomized and randomly received in the experimental arm gemcitabine 1000 mg/m2 on days 1 and 15 and cisplatin 25 mg/m2 with RHT on days 2, 3 and 16, 17 (n= 57). In the control arm, gemcitabine 1000 mg/m2 was given on days 1, 8, and 15 (n=60). The adjuvant therapy was administered every four weeks and patients received six treatment cycles. The trial was prematurely stopped in May 2018, when the modified FOLFIRINOX regimen replaced gemcitabine as standard therapy.
While the HEAT trial did not demonstrate a significant difference in disease-free survival (12.7 vs. 11.2 months; p = 0.394), the results show a significant improvement in post-recurrence survival in the experimental arm (15.3 vs. 9.8 months; p = 0.031). In case of recurrence m-FOLFIRINOX or nab-paclitaxel plus gemcitabin was used in most patients, which was not different between the trial arms. In addition, a trend for improved overall survival of 33.2 vs. 25.2 months (p = 0.099) was observed, with 5-year survival rates of 28.4% vs. 18.7%, respectively. Adverse incidents according to CTCAE grade ≥ 3 did not differ significantly between groups (61.5% vs. 63.6%) and toxicity was lower than with intensified adjuvant chemotherapy with modified FOLFIRINOX.

Reference:
Refractory or recurrent non-testicular malignant germ-cell tumors in children and adolescents
Paediatric malignant germ-cell tumors are rare, accounting for about 3–4% of all malignancies in children and more than 50% are diagnosed in infants and toddlers younger than 4 years1. Cisplatin based polychemotherapy leads to cure rates of up to 90% of patients, however around 10% of patients have refractory or recurrent disease.
An open-label, non-randomized, single-institution, phase 2 study tested the combined treatment with 3-4 cycles of cisplatinum, etoposide and ifosfamide chemotherapy (all applied day 1-4) combined with regional hyperthermia (day 1 and 4 of every cycle, 60 minutes 42°C) in 44 patients with refractory or recurrent paediatric malignant germ-cell tumors1. Of all patients 86% had an objective response to treatment. 5-year event-free survival was 62% (95% CI 45–75), and 5-year overall survival was 72% (95% CI 55–83)1. The long-term prognosis of patients with poor response or after first relapse was almost similar to those patients receiving first-line treatment, thus the data of this trial suggests that hyperthermia may serve to advance chemoresistence1.
Reference:
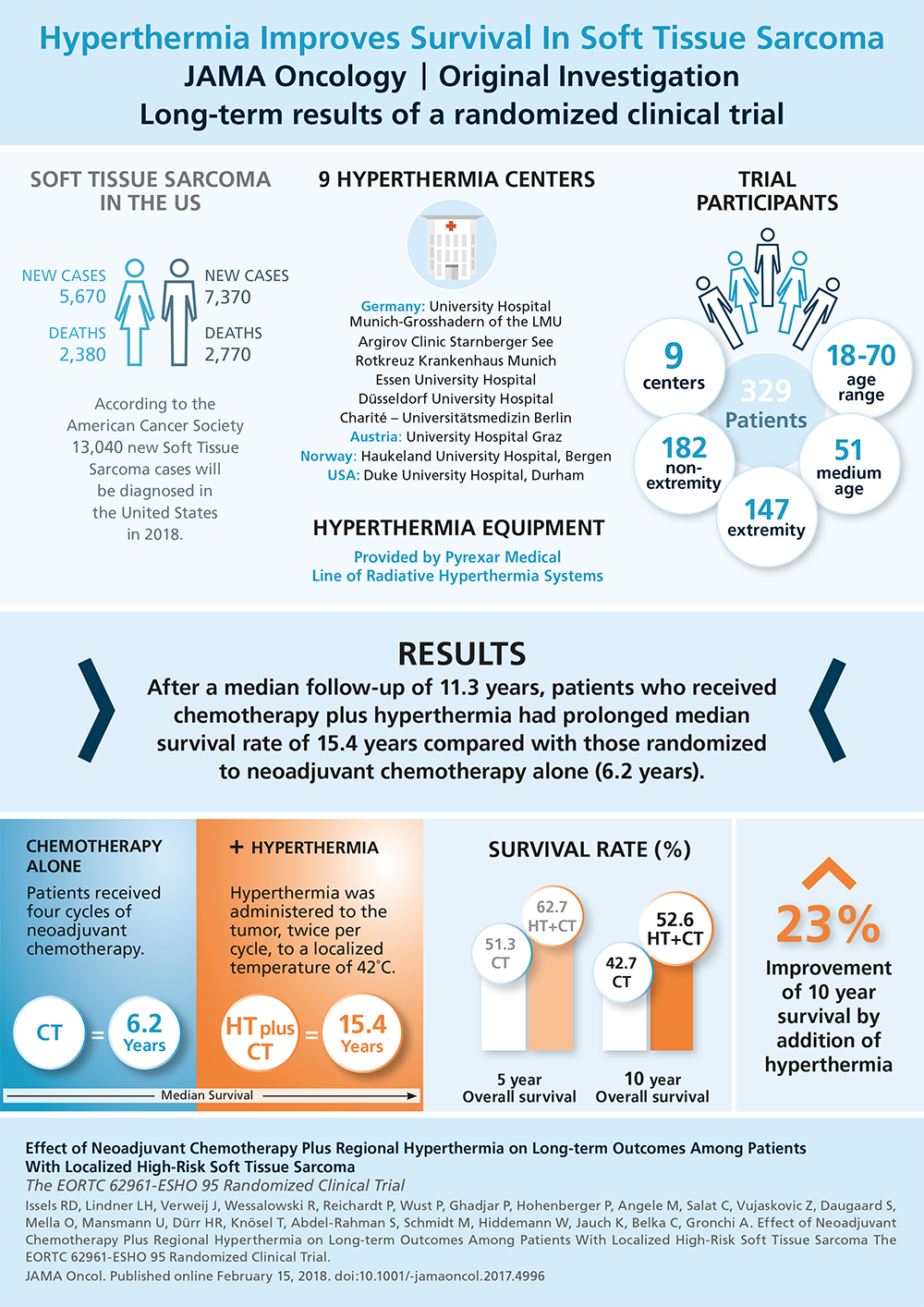
High-risk soft tissue sarcoma
Soft tissue sarcoma is a relatively rare disease accounting for less than 1% of all cancer cases. Surgery and neoadjuvant or adjuvant radiation therapy represents the standard of care for high-risk soft tissue sarcoma, perioperative chemotherapy is an additional treatment option.
Long term results of a randomized trial are available comparing perioperative chemotherapy (using doxorubicin, ifosfamide, and etoposide, 4 cycles neoadjuvant, 4 cycles adjuvant) plus regional hyperthermia vs. perioperative chemotherapy alone in 341 patients with primary or locally recurrent high-risk soft tissue sarcoma of the trunk and the extremities1, 2. Adjuvant radiation therapy was given with 50-66 Gy and there was no difference between the two arms with respect to the utilization of radiation therapy. Hyperthermia (42°C for a 60-minute period) was given concurrently with ifosfamide on day 1 and day 4 of each chemotherapy cycle. Compared with chemotherapy alone, adding regional hyperthermia improved the primary endpoint of this trial being local progression-free survival (hazard ratio, 0.65; 95% CI, 0.49-0.86; P = .002). Furthermore, patients randomized to chemotherapy plus regional hyperthermia had prolonged overall survival rates compared with those randomized to chemotherapy alone (hazard ratio, 0.73; 95% CI, 0.54-0.98; P = .04) with 5-year survivalof 62.7% (95% CI, 55.2%-70.1%) vs. 51.3% (95% CI, 43.7%-59.0%), respectively, and 10-year survival of 52.6% (95% CI, 44.7%-60.6%) vs 42.7% (95% CI, 35.0%-50.4%)2.
References: